Tungsten Trioxide Fuel Cell Carrier
Tungsten trioxide fuel cell carrier working environment has the characteristics of high voltage, high moisture and low pH value, so the electrode material is required to have anti corrosion property. The traditional fuel cell catalyst is using carbon as fuel cell carrier to carry Pt. However, the weak anti corrosion property of carbon lowers the catalytic property of catalyst which shorter the service life.
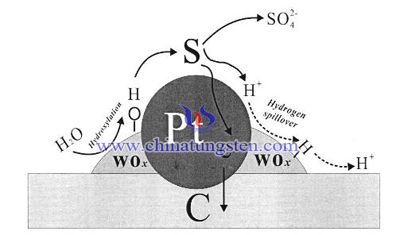
Tungsten trioxide (WO3) has stable chemical property and strong acid resistance, it is suitable for fuel cell reaction. Research shows that WO3 can be used to replace carbon as fuel cell carrier to carry precious metal and form Pt/WO3/C catalyst. It can promote anti corrosion property of catalyst and prolong its service life. Besides that, due to the concerted catalysis of WO3 and Pt can lower the cost and reduce the usage of precious metal, catalytic property is also strengthened. In the leaching test of CO in WO3 catalyst, Pt.WO3/C shows good CO oxidation ability, it means WO3 fuel cell carrier has high performance of anti CO poisoning property.
WO3/Pt/C Fuel Cell Carrier Anti-Sulfur Poisoning Property
Surface of WO3 has well hydrophilia, it can be easily reduced to form mixing oxides which contains hydroxy on the surface with valence +5 and +6. It can accelerate oxidation reaction between electron and surface absorption material so as to promote electron transition rate between WO3 and Pt. Sox addicted on the surface of catalyst will oxidize faster and desorbed from the surface, SOx poisoning effect is lowered, fuel cell property is gradually recovered then. Apart from that, WO3 can react with active hydrogen ion from Pt catalyst, tungsten bronze is formed. WO3 as fuel cell carrier can also promote methanoic acid property. WO3-Pt/C catalyst with proper mass ratio can extremely lower toxic of SO2 and speed its property recovery ratio, its catalytic property and stability also get better.
If you have any interest in tungsten trioxide, please feel free to contact us by email: sales@chinatungsten.com or by telephone: +86 592 5129696.
More info>>
