Pyrochlore Type Tungsten Oxide
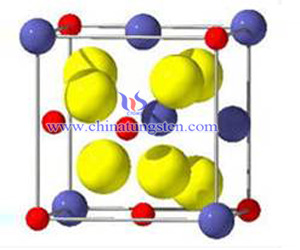
Chemical formula of pyrochlore type tungsten oxide is H2W2O7 Or WO3·0.5H2O。
Structure
Its atomic structure motifs are distorted octahedral WO6, and form a layered structure with six-membered ring channels through the angular of rigid skeleton tungsten-oxide, and has a three-dimensional pore along the (111) direction.
Synthesis Approach
There is a variety of pyrochlore tungsten oxide synthesis approaches, within that two ways are the most important:
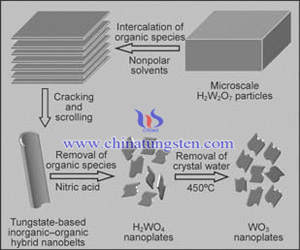
1.Carry out acid treatment on the precursor of Aurivillius-type compound of Bi2W2O9 to give the pyrochlore type tungsten oxide. Detailed process is like: mixing the Bi2O3 and WO3 in accordance with 1:2 molar ratio uniformly, and then calcining under 800℃ for 48 hours to generate Bi2W2O9 and put it into concentrated hydrochloric acid, followed by vigorous stirring at room temperature for 72h to obtain undistorted crystals of H 2 W2O7.
2.Hydrothermal synthesis method: carry out hydrothermal synthesis to a certain time after acidified the pH value of sodium tungstate solution to a certain range.
(Note: pH value range still controversial, according to studies of different scientists, pyrochlore tungsten oxide can be prepared when pH value is greater than 3.5 and even the alkaline range).
Application
Because pyrochlore type tungsten oxide has the mesh structure and dielectric stability like zeolite, it has a wide range of applications in the fields of material science; at the same time, due to it can be used as intermediates for the preparation of tungsten oxide in tungsten smelting, it is suggested to hopefully cut down the process to achieve lye circulation; in addition, it also has good prospects in layered composite materials, sensors and other fields.
If you have any interest in tungsten trioxide, please feel free to contact us by email: sales@chinatungsten.com or by telephone: +86 592 5129696.
More info>>
